FDA: Valsartan Tablets recalled
Federal officials say a pharmaceutical company is recalling a heart medication as one of its chemicals could possibly cause cancer.
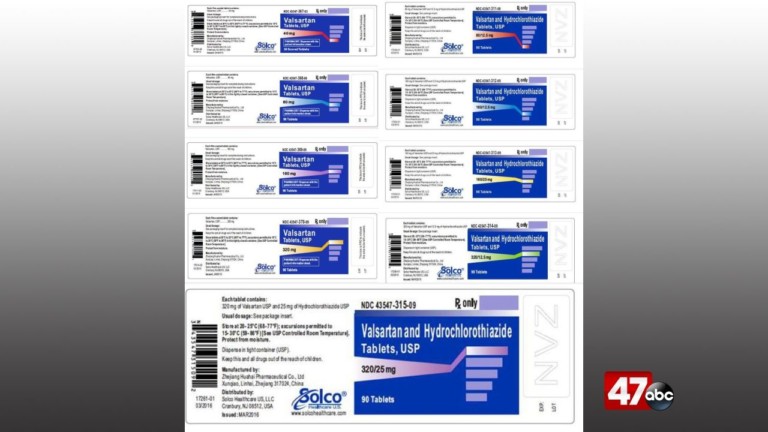
The United States Food and Drug Administration says Prinston Pharmaceutical Inc. and Solco Healthcare LLC. is recalling Valsartan tablets (40 mg, 80mg, 160mg, and 320mg) and Valsartan-Hydrochlorothiazide tablets (80mg/12.5mg, 160mg/12.5mg, 160mg/25mg, 320mg/12.5mg, and 320mg/25mg) to the retail level. Officials say the recall comes after a trace amount an unexpected impurity, N-nitrosodimethylamine (NDMA) was detected.
Authorities say the impurity has been classified as a probable human carcinogen as per International Agency for Research on Cancer (IARC) classification.
FDA says the products are re indicated for the treatment of hypertension.
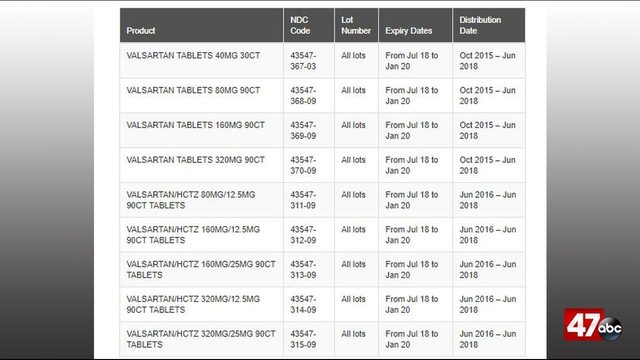
Below is the product information for the recalled products:
Officials report that Solco Healthcare LLC. is notifying distributors and consumers by letter and email arranging for return of the recall product.
For more information regarding the recall, call Solco Customer Service at 1-866-931-9829 or email customerservice@solcohealthcare.com.


